Topic outline
General
 General Information
General Information Memorize!
The following documents contain information to memorize for the course. We will be using the information all year, so it is imperative that you begin study as soon as possible. The dates in the titles are the tentative days for the quiz.
Topic 9
 Fish Kill—Finding the Solution: Instructions
Fish Kill—Finding the Solution: InstructionsYou and your classmates will participate in a Riverwood Town Council Meeting in which special interest groups will voice their position on the fish kill issue and the town council will decide who is responsible and who must pay. Some members of your class will moderate the “Fish Kill—Who Pays?” Others will represent the various special interest groups outlined in the book. Please Note: This is a role-modeling activity! Get into your roles! Use acting, costumes, banners, slogans, etc.
Toxic Waters: A series about the worsening pollution in America's waters and regulators' response.
-A series of articles from the New York Times
Class Resources
Forums for each Special Interest Group
Water Testing Results
Newspaper Articles About Riverwood Fish Kill --These are the articles used for Assignment #1 and other articles relating to water in Riverwood.
Upload Presentations for Town Council Meeting using the link below.
Due Wednesday at the end of the class period.
Topic 11
Unit 5: Covalent Bonding- 4. I can recognize ionic compounds, covalent compounds, and acids by name and formula.
- 5. I can name covalent compounds.
- 2. I can draw Lewis structures for single, double, and triple covalent bonds.
- 6. I can write the formula for covalent compounds.
- 7. I can write the formula for ionic compounds.
More online practice naming compounds.
You can choose ionic, molecular (covalent), or both.
Topic 12
Unit 4--Ionic, Covalent & Metallic Bonding

11. I can use the periodic table to predict the type of bond formed between elements (ionic, covalent, or metallic)
8. I can explain the general trends in physical properties of ionic compounds: melting point, boiling point, hardness and conductivity.
2. I can generalize about the strength of ionic bonds based on the physical properties of ionic compounds.
3. I can name ionic compounds.
4. I can write the chemical formulae for ionic compounds.
6. I can describe a metallic bond.
7. I can use the electron sea model of electrons to explain the physical properties of metals: luster, malleability, conductivity, melting point, & boiling point.
8. I can define and give examples of alloys.
9. I can name and write the formula for acids.
Scroll to bottom of page for practice and answer key.
8. I can distinguish between binary acids and oxyacids


Topic 13
Introduction to the Mole
- 1. I can explain how a mole is used to indirectly count the number of particles in matter.
- 2. I can convert between moles and number of particles.
- 3. I can calculate molar mass.
- 4. can convert between the mass of an element and the number of particles.
- 5. I can convert between mass and the number of moles.
- 6. I can explain what is meant by the percent composition of a compound and calculate percent composition.
- 7. I can define and give examples of empirical formulas and molecular formulas.
- 8. I can determine the empirical formula from percent composition and from mass data.
- 9. I can determine a molecular formula from percent composition.
- 10. I can explain what a hydrate is and relate the name of the hydrate to its composition.
Topic 14
Chemical Equations--Balancing and Identifying Types
1. I can recognize evidence of chemical change.
2. I can represent chemical reactions with equations.
3. I can balance chemical equations.
4. I can classify chemical reactions: synthesis, combustion, single replacement, double replacement and decomposition.
5. I can use an activity series to predict whether or not single replacement reactions will occur.
6. I can describe aqueous solutions.
7. I can predict the products of chemical reactions and write their formula.
a. I can predict whether reactions in aqueous solutions will produce a precipitate, water, or gas.
8. I can write complete ionic and net ionic equations for chemical reactions in aqueous solutions.
Lab Videos
Test Review Materials
Topic 16
Stoichiometry
Chapter 11
1. I can calculate the mass of a particular substance formed from the masses of starting materials. (Stoichiometry)
Practice Problems: p. 392-397 #60-62, 64-66
2. I can identify the limiting reactant in a chemical equation.
Practice Problems: p. 392-397 #74, 75, 79, 82,
3. I can identify and calculate the amount of reactant in excess.
Practice Problems: p. 392-397 #82, 93
4. I can calculate the mass of the products (theoretical yield).
Practice Problems: p. 392-397 #80-82, 90-93
5. I can calculate the percent yield of a chemical reaction.
Practice Problems: p. 392-397 #83, 84, 90, 91, 102
Lab Materials
Topic 17
Bonding & States of Matter
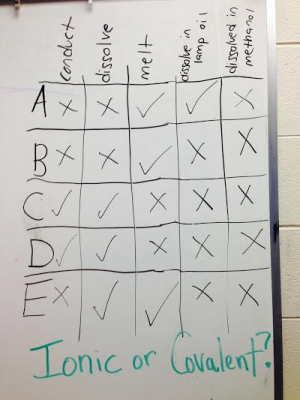
I can use the periodic table to predict the type of bond formed between elements (ionic, covalent, or metallic)
I can explain the general trends in physical properties of ionic compounds: melting point, boiling point, hardness and conductivity.
#1. I can draw Lewis structures for atoms and molecules.
#2. I can draw modified structures for molecules showing the bonds and bond dipoles (if they exist).
3. I can identify the shape of a molecule.
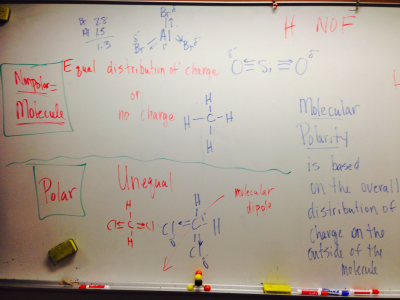
#4. I can identify if a bond is polar or nonpolar using the difference in electro-negativities of the atoms.
#5. I can determine if a molecule is polar or nonpolar based on the molecule’s shape and the polarity of its bonds.
#6. I can identify the elements necessary for hydrogen bonds to form between molecules.
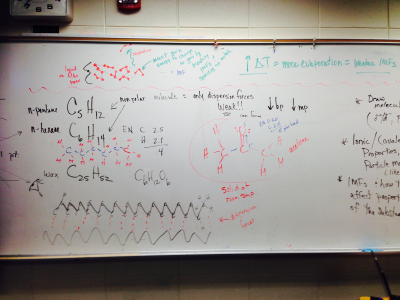
#7. Given a structural formula, I can identify the type of intermolecular forces present (dispersion, dipole-dipole, or hydrogen)
#8. I can predict if the forces in a solid are primarily metallic, covalent, network covalent, or ionic based on the element’s location on the periodic table.
#9. I can explain the properties of various solids such as malleability, conductivity, melting points, etc. in terms of the solid’s structure, IMFs (intermolecular forces), and bonding.
#10. I can explain why both the melting points and the boiling points for water are significantly higher than other molecules of comparable mass (ex. Methane and ammonia)
- This topic
Fresh Juice Research
We will use this section to share background information, explain experimental designs, and discuss our research.
Use this space to add research, experimental design, and data. You can comment on the contributions of others.